Rapid antibody development is urgently needed to tackle outbreaks of emerging infectious diseases. Creating therapeutics for these disease-causing agents historically required years of clinical and regulatory assessment before they become available to patients.
The Sasisekharan Lab at MIT and SMART, Singapore, has been actively exploring ways to condense drug development timelines by performing many of the necessary steps in parallel, in contrast to the traditional linear experimental approach to drug development.
Sasisekharan Lab’s approach considers the regulatory framework as the driving design constraint. The Lab redesigns the traditional linear sequential process to run experiments in parallel. With this approach and the Antibody Engineering methods, and by working with regulators each step of the way, Sasisekharan Lab has demonstrated that it can rapidly develop, produce, and evaluate antibody candidates for safety and efficacy in humans.
This framework, with advanced instrumentation support from Agilent Technologies, has led to biologics development (including GMP manufacturing and non-clinical studies) and first-in-human studies with unprecedented timelines.
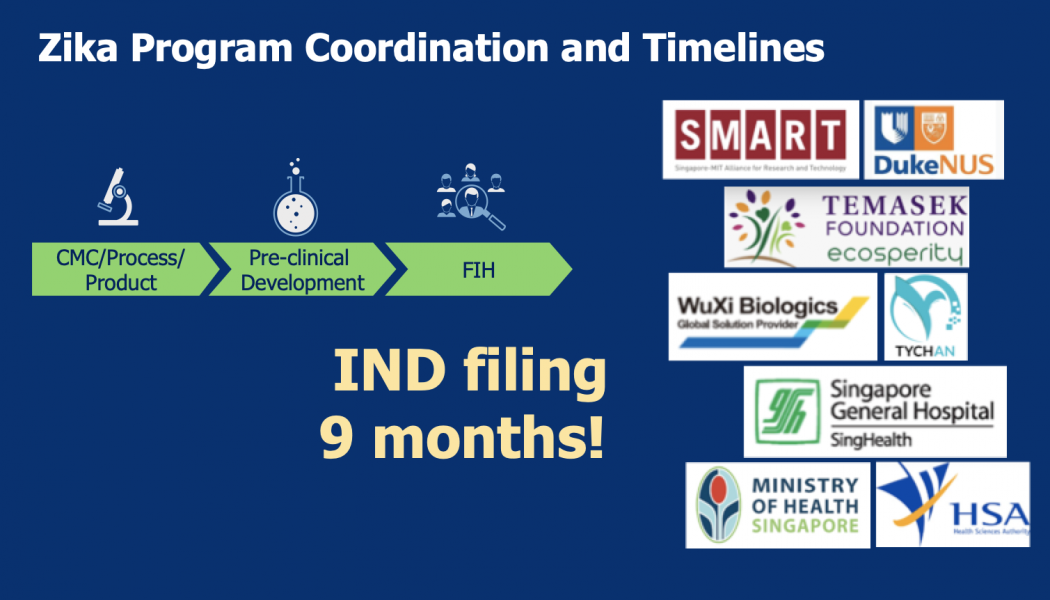
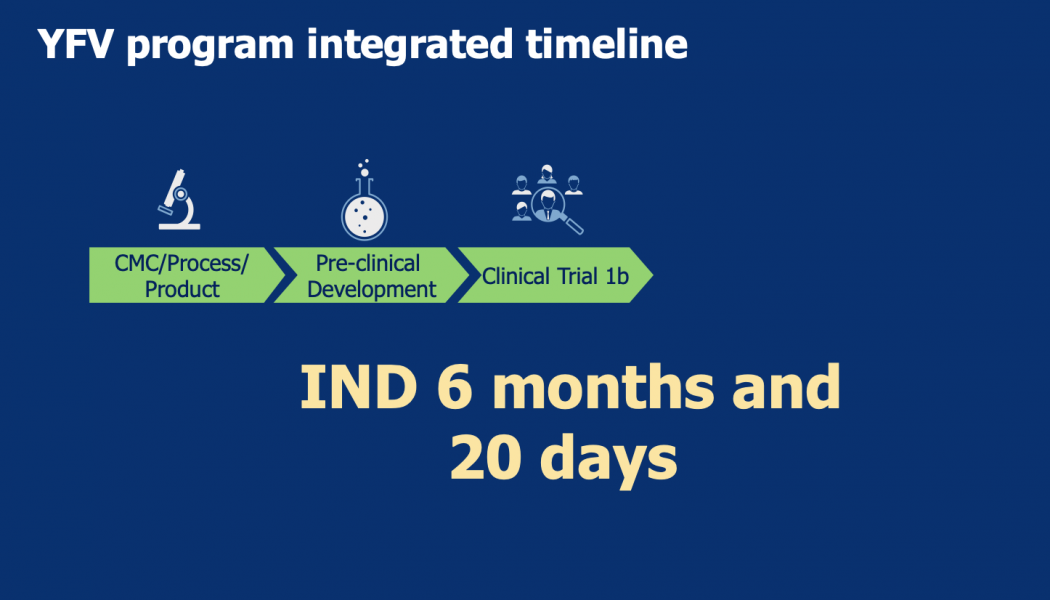
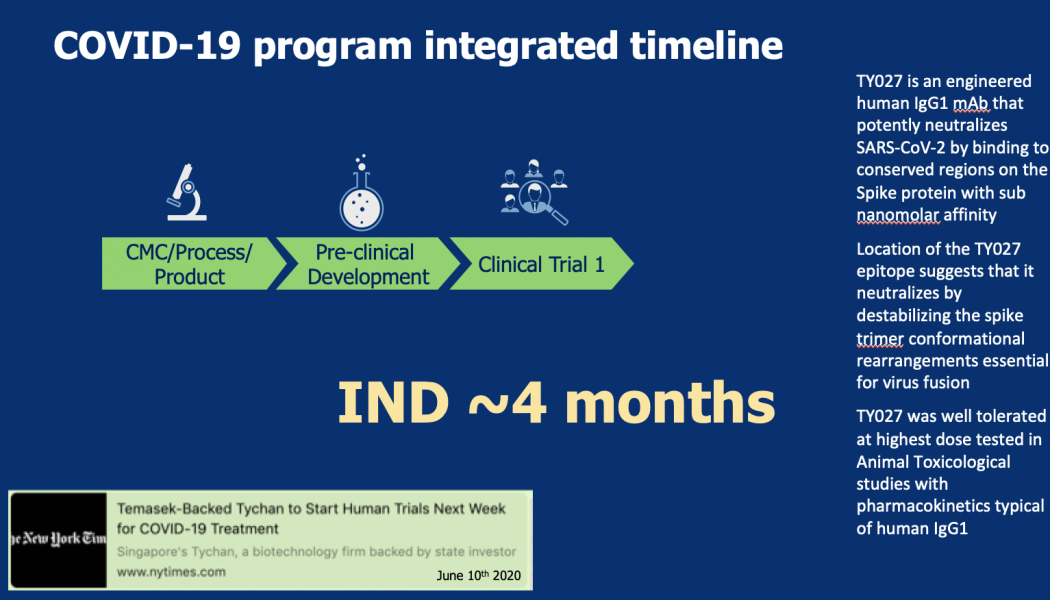
Sasisekharan Lab and partners moved from design to first-in-human infusion in nine months for Zika virus, about seven months for Yellow Fever virus, and most recently four months for SARS-CoV-2 (COVID 19)
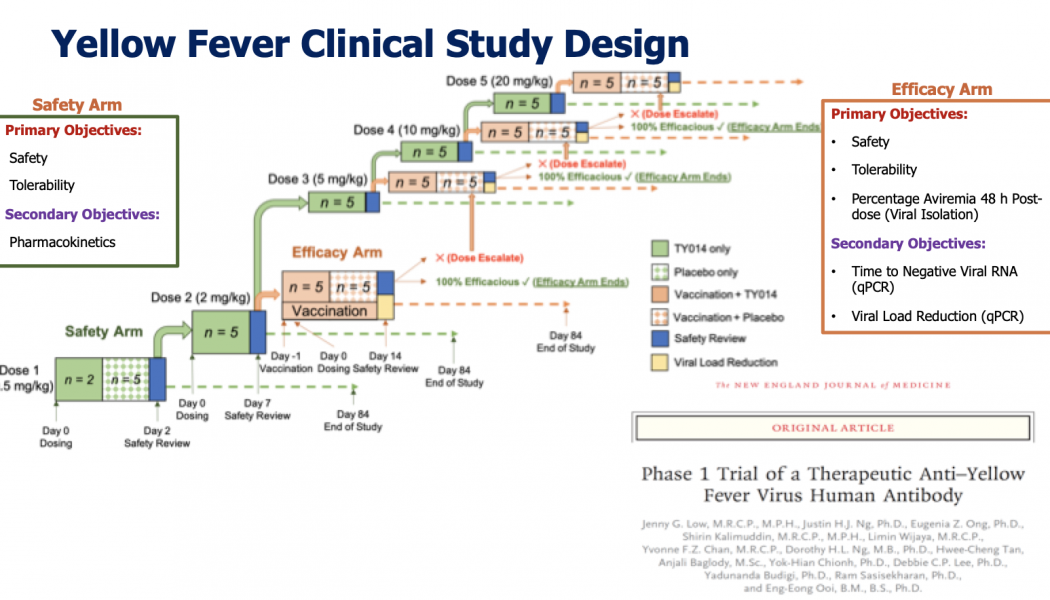
Rapid antibody development yields possible treatment for yellow fever
Hastening vaccines and treatments
please click forward arrow to see the slides